M.W. Little, K.K. Murray, “Two-Laser Mid-Infrared and Ultraviolet Matrix-Assisted Laser Desorption/Ionization,” Int. J. Mass Spectrom. 261 (2007) 140–145. doi:10.1016/j.ijms.2006.08.010.
Abstract

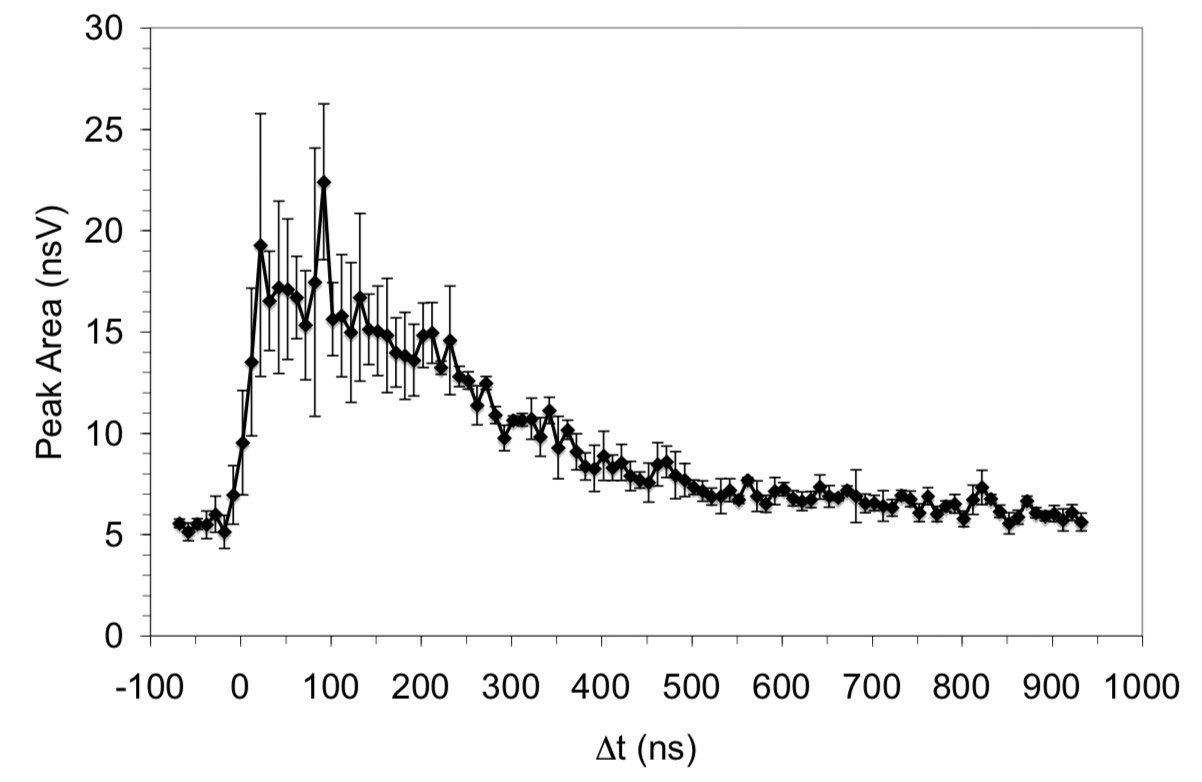
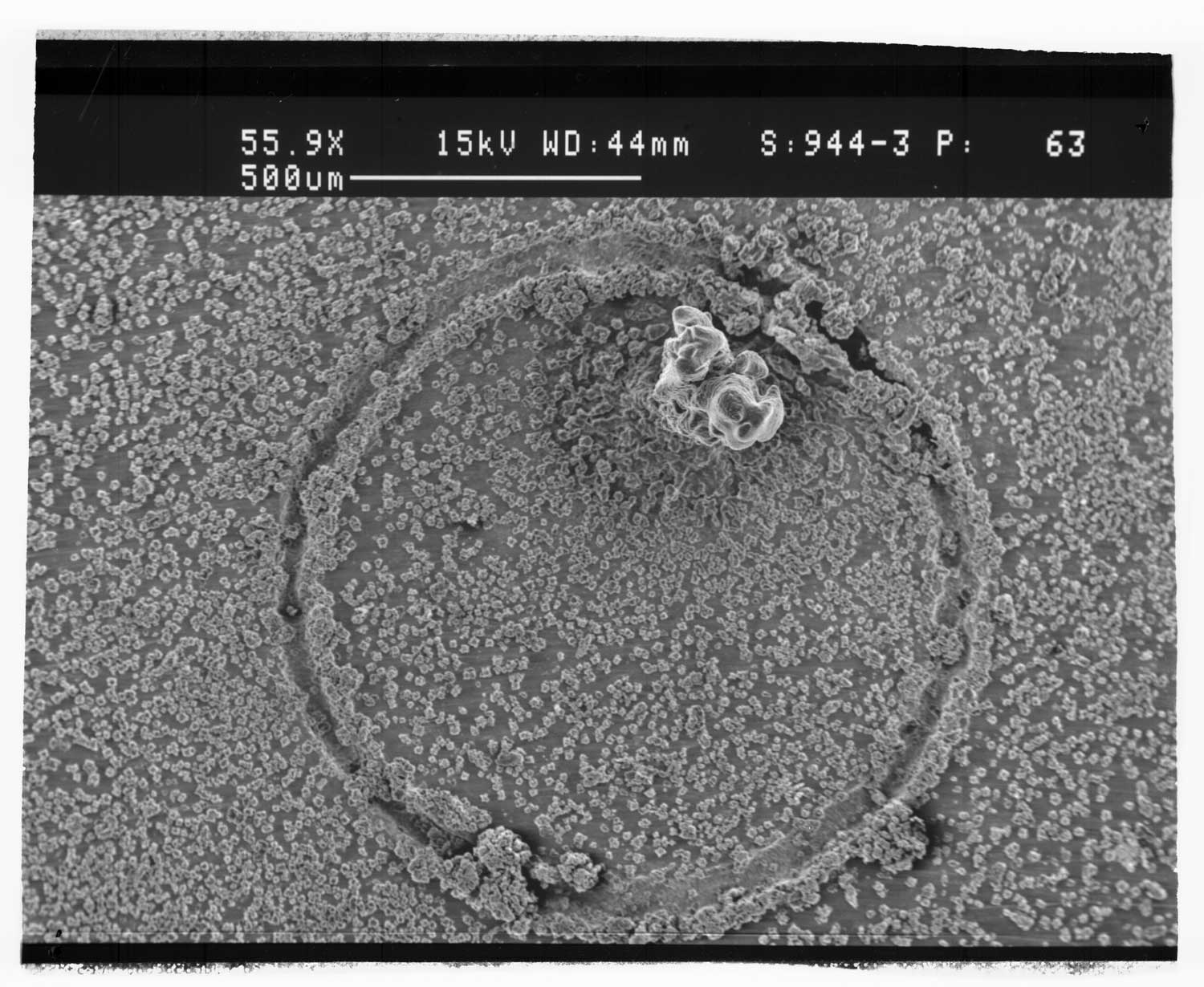
Matrix-assisted laser desorption/ionization (MALDI) was performed using two-pulsed lasers with wavelengths in the infrared and ultraviolet regions. A 2.94 μm pulsed optical parametric oscillator laser system and a 337 nm pulsed nitrogen laser irradiated the same spot on the sample target. Sinapinic acid (SA), 2,5-dihydroxybenzoic acid (DHB), α-cyano-4-hydroxycinnamic acid (CCA), and 4-nitroaniline (NA) were used as matrices, and bovine insulin and cytochrome C were used as analytes. The laser energy was adjusted so that one-laser MALDI and LDI was at a minimum and the matrix and analyte ion signal was enhanced when the two lasers were fired together. Two-laser LDI was observed with SA, DHB, and NA matrices and two-laser MALDI was observed with SA and DHB. Plots of ion signal as a function of delay between the IR and UV lasers show two-laser signal from 0 ns up to a delay of 500 ns when the IR laser is fired before the UV laser. The results are interpreted in terms of IR laser heating of the target that leads to an enhancement in UV LDI and MALDI.